Each of the two unpaired electrons of the oxygen atom will form a bond with one of the unpaired electrons of the hydrogen atoms. With few exceptions, hydrogen atoms are always placed on the outside of the molecule, and in this case the central atom would be oxygen. We can now see that we have eight valence electrons (six from oxygen and one from each hydrogen). Water has the chemical formula of H 2O, which means there The Lewis structure of each of these atoms would be as follows: After adding the four unshared electrons around element symbol, form electron pairs using the remaining two outer shell electrons.Īre two hydrogen atoms and one oxygen atom. If the electrons are not placed correctly, one could think that oxygen has three lone pairs (which would not leave any unshared electrons to form chemical bonds). For example, oxygen has 6 electrons in the outer shell, which are the pattern of two lone pairs and two singles.
LEWIS STRUCTURE GENERATOR ONLINE HOW TO
How to Build a Lewis Structure?įor simple molecules, the most effective way to get the correct Lewis structure is to write the Lewis diagrams for all the atoms involved in the bonding and adding up the total number of valence electrons that are available for bonding. Lewis structures display the electrons of the outer shells because these are the ones that participate in making chemical bonds.
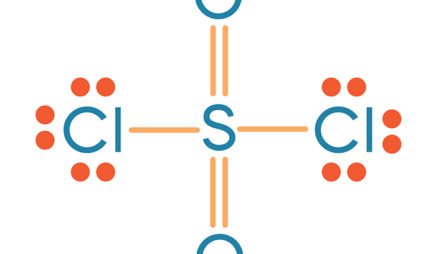
The octet rule states that an atom in a molecule will be stable when there are eight electrons in its outer shell (with the exception of hydrogen, in which the outer shell is satisfied with two electrons). The atoms in a Lewis structure tend to share electrons so that each atom has eight electrons (the octet rule). Most bonding is not purely covalent, but is polar covalent (unequal sharing) based on electronegativity differences. Covalent bonds are stronger than the electrostatic interactions of ionic bonds, but keep in mind that we are not considering ionic compounds as we go through this chapter. Covalent bonds are a type of chemical bonding formed by the sharing of electrons in the valence shells of the atoms. The Lewis structure is used to represent the covalent bonding of a molecule or ion. These diagrams are based on the electron structures learned in the Atomic Structure and Periodic Table chapters. Where the symbol represents the element (in this case, hydrogen, helium, and carbon) and the dots represent the electrons in the outer shell (in this case, one, two, and four). For example, the Lewis diagrams for hydrogen, helium, and carbon are Lewis diagrams, also called electron-dot diagrams, are used to represent paired and unpaired valence (outer shell) electrons in an atom. Lewis Structures for Electron-poor Compounds.Lewis Structures for Electron-rich Compounds.

Lewis Structures and Resonance Structures.
